PSA, Total
Main Content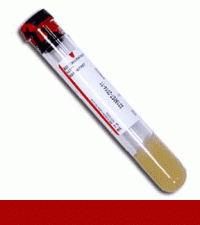
Test Code: 5363
CPT Code(s): 84153
Methodology: Immunoassay (IA)
Limitations: Based on solid evidence, screening with PSA and/or DRE detects some prostate cancers that would never have caused important clinical problems. Current prostate cancer treatments, including radical prostatectomy and radiation therapy, result in permanent side effects in many men. The most common of these side effects are erectile dystunction and urinary incontinence. Whatever the screening modality, the screening process itself can lead to adverse psychological effects in men who have a prostate biopsy but do not have identified prostate cancer. Prostatic biopsies are associated with complications, including fever, pain, hematospermia/hematuria, positive urine cultures, and rarely sepsis. Lower PSA levels are associated with obesity.
Clinical Significance: Elevated serum PSA concentrations have been reported in men with prostate cancer, benign prostatic hypertrophy, and inflammatory conditions of the prostate.
Additional names: Prostate Specific Antigen
Supply: #T01 - Red/Gray SST 8.5mL
Preferred Specimen Requirements: Serum
Preferred Volume: 1mL
Container Type: Serum Separator tube (SST)
Special Instructions: Do not collect specimen after a transrectal biopsy, results may be falsely elevated.
Transport Temperature: Room Temperature
Specimen Stability: Room Temperature: 7 days
Reject due: Specimens exceeding stability; Unlabeled or improperly labeled specimens; Specimens other than serum
For additional supply or collection device information, please contact DLO's Customer Service at (800) 891-2917, option 2.
The CPT codes provided are based on AMA guidelines and are for informational purposes only. CPT coding is the sole responsibility of the billing party. Please direct any questions regarding coding to the payor being billed.


CLIA