Chlamydia / N. Gonorrhoeae RNA, TMA - Vaginal Collection
Main Content
Test Code: 11363
Includes: Chlamydia trachomatis, Neisseria gonorrhoeae
Methodology: Dual Kinetic Assay (DKA) • Target Capture • Transcription-Mediated Amplification (TMA)
Reference Range(s):
C.trachomatis RNA, TMA - Not detected
N.gonorrhoeae RNA, TMA - Not detected
This test was performed using the APTIMA® COMBO2 Assay (GEN-PROBE).
Clinical Significance:
C. trachomatis infections are the leading cause of sexually transmitted diseases in the United States. C. trachomatis is known to cause cervicitis, pelvic inflammatory disease (PID), epididymitis and proctitis. It is also the most frequent cause of non-gonococcal urethritis in men. Among women, the consequences of Chlamydial infections are severe if left untreated. Approximately half of Chlamydial infections are asymptomatic.Neisseria gonorrhoeae (gonococci) is the causative agent of gonorrhea. In men, this disease generally results in anterior urethritis accompanied by purulent exudate. In women, the disease is most often found in the cervix, but the vagina and uterus may also be infected.
Test FAQ: Chlamydia trachomatis and Neisseria gonorrhoeae RNA, TMA
Alternative Name(s): CT/NG APTIMA, CT/GC APTIMA, Hologic, CT/GC TMA, CT/NG TMA
Additional Preferred Specimens Collection for Test Code 11363:
1 ml liquid cytology collected through Thinprep® Broom or Brush/Spatula
0.5 ml of Surepath® preservative fluid collected using Broom or Brush/Spatula
2 ml urine using APTIMA Urine Specimen Collection Kit
Endocervical or Urethral Swabs in APTIMA Combo 2 Assay Unisex Swab Specimen Collection Kit
Note: Results from the APTIMA Combo 2 Assay should be interpreted in conjunction with other laboratory and clinical data available to the clinician.
The APTIMA Combo 2 Assay is not intended for the evaluation of suspected sexual abuse or for other medico-legal indications.
For a complete list of assay limitations we recommend that you review the manufacturers package insert, http://www.hologic.com/package-inserts/clinical-diagnostics- blood-screening/assays-and-tests/aptima-combo-2-assay-ctng.
APTIMA Vaginal Swab Device Collection Guide
Supply: A03 - APTIMA Vaginal Collection
Test Code:
11363, CT/NG
19550(X), SureSwab Trichomonas vaginalis
10121(X), SureSwab Advanced Cadida Vaginitis (CV), TMA
10016(X), SureSwab Advanced Bacterial Vaginosis (BV), TMA
10120(X), SureSwab Advanced Vaginitis Plus, TMA
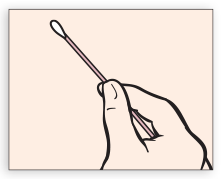
Partially open swab package. Do not touch the soft tip or lay swab down. If the soft tip is touched, laid down, or dropped, discard and get a new APTIMA Vaginal Swab Specimen Collection Kit. Hold swab, placing thumb and forefinger in the middle of the swab shaft. Do not hold the swab shaft below the score line.
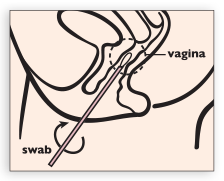
Carefully insert swab into vagina about 2 inches (5 cm) past the introitus and gently rotate the swab for 10 to 30 seconds. Make sure the swab touches the vagina walls so that moisture is absorbed by the swab and withdraw swab without touching the skin.
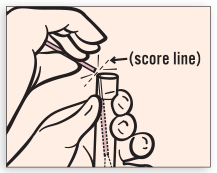
While holding swab in hand, unscrew the tube cap. Do not spill tube contents. If the tube contents are spilled, discard and get a new APTIMA Vaginal Swab Specimen Collection Kit. Immediately place swab into transport tube so that the tip of the swab is visible below tube label. Carefully break swab shaft at the score line against the side of the tube.
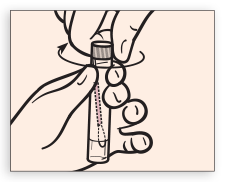
Tightly screw cap onto tube.
Transport container: APTIMA® transport tube
Transport temperature: Room temperature
Cervical/vaginal swabs in APTIMA® transport:
Room temperature: 60 days
Refrigerated: 60 days
Frozen: 1 year
For additional supply or collection device information, please contact DLO's Customer Services at (800) 891-2917, option 2.


CLIA