Respiratory Syncytial Virus (RSV), Immunoassay
Test code: 8467
Methodology: Immunochromatography
Includes: Respiratory syncytial virus (RSV)
Alternative name(s): RSV, RSV AG by EIA, RSV EIA, RSV-Direct Antigen
Test code: 8467
Methodology: Immunochromatography
Includes: Respiratory syncytial virus (RSV)
Alternative name(s): RSV, RSV AG by EIA, RSV EIA, RSV-Direct Antigen
Test Code: 95045
Pathogens detected: Adenovirus, Norovirus, Rotavirus, Enterohemorrhagic E. Coli, Shiga-like Toxin, Enteropathogenic E. Coli, Enterotoxigenic E. Coli, Enteroinvasive E. Coli/Shigella, Salmonella Enterica, Campylobacter Jejuni, Vibrio Parahaemolyticus, Cdiff Toxin B Gene, Cryptosporidium Parvum, Giardia Lamblia
Methodlogy: TEM-PCR
Clinical Significance: Rapid target enriched multiplex molecular testing of the presence of common gastrointestinal pathogens
Test code: 15059
Methodology: Centrifugation enhanced culture using R-mix cell line
Includes: Respiratory syncytial virus (RSV)
Alternative name(s): RSV Rapid culture, Respiratory Syncytial Virus Rapid Culture
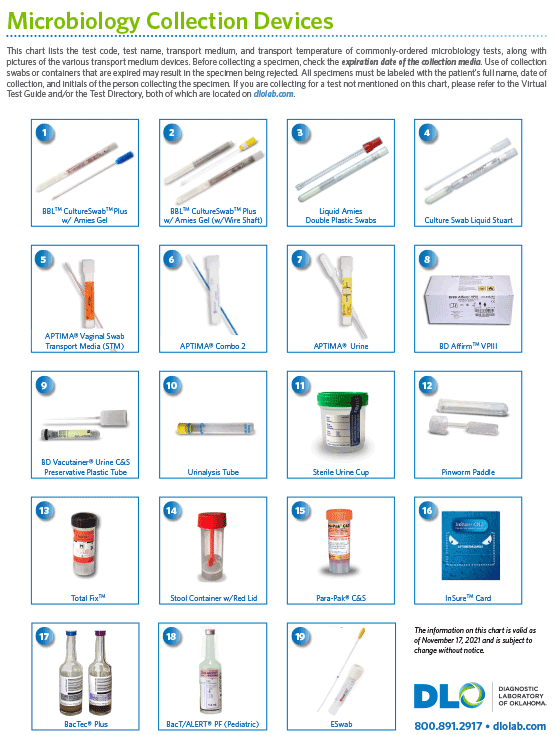
For additional supply or collection device information, please contact DLO's Customer Services at (800) 891-2917, option 2.
Test Code: 4550
CPT Code(s): 87070
Test code: 11177
CPT code(s): 87804 (x2)
Methodology: Optical Immunoassay
Includes: Influenza A & B
Alternative name(s): Influenza A&B EIA; Rapid Flu; Rapid Influenza
Test Code: 36970
Methodology: Immunoassay (IA)
Clinical Significance: QuantiFERON®-TB Gold IT is an indirect test for M. tuberculosis infection (including disease) and is intended for use in conjunction with risk assessment, radiography and other medical and diagnostic evaluations.
Alternative Name(s): TB Gold, QFT Gold Plus, QFT TB, QFT Plus, IGRA
Test Code: 10306
CPT Code(s): 80074
Methodology: See individual test
Includes: Hepatitis A IgM Antibody; Hepatitis B Surface Antigen with Reflex to Confirmation; Hepatitis B Core Antibody (IgM); Hepatitis C Antibody with Reflex to HCV, RNA, Quantitative, Real-Time PCR; Hepatitis B Surface Antigen: Positive samples will be confirmed based on the manufacturer's FDA approved recommendations at an additional charge (CPT code(s): 87341).
Test Code: 6462
CPT Code(s): 86704, 86706, 86708, 86803, 87340
Methodology: See individual test
Includes: Hepatitis A Antibody, Total; Hepatitis B Surface Antibody, Qualitative; Hepatitis B Surface Antigen with Reflex Confirmation; Hepatitis B Core Antibody, Total; Hepatitis C Antibody with Reflex to HCV, RNA, Quantitative, Real-Time PCR
Test Code: 8472
CPT Code(s): 86803
Methodology: Immunoassay (IA)
Includes: If Hepatitis C Antibody is reactive, then Hepatitis C Viral RNA, Quantitative, Real-Time PCR will be performed at an additional charge (CPT code(s): 87522).